Discussion
schmunk said:
K12beano said:
tr7v8 said:
The roundest knight at King Arthur’s round table was Sir Circumference. He acquired his size from eating too much pi.
...and often linked to a girl, Di Amater S'phia
marshalla said:
schmunk said:
K12beano said:
tr7v8 said:
The roundest knight at King Arthur’s round table was Sir Circumference. He acquired his size from eating too much pi.
...and often linked to a girl, Di Amater S'phia
Brother D said:
What do they teach in GCSE and ALevel physics/chemistry nowadays in the UK?
At school it was just discrete shells each containing 2,8 etc. (Probably mistaken, but even at uni I don't recall my girlfriend at the time studying phys and molecular chem referring to orbital model (this was 20 years ago thou)?
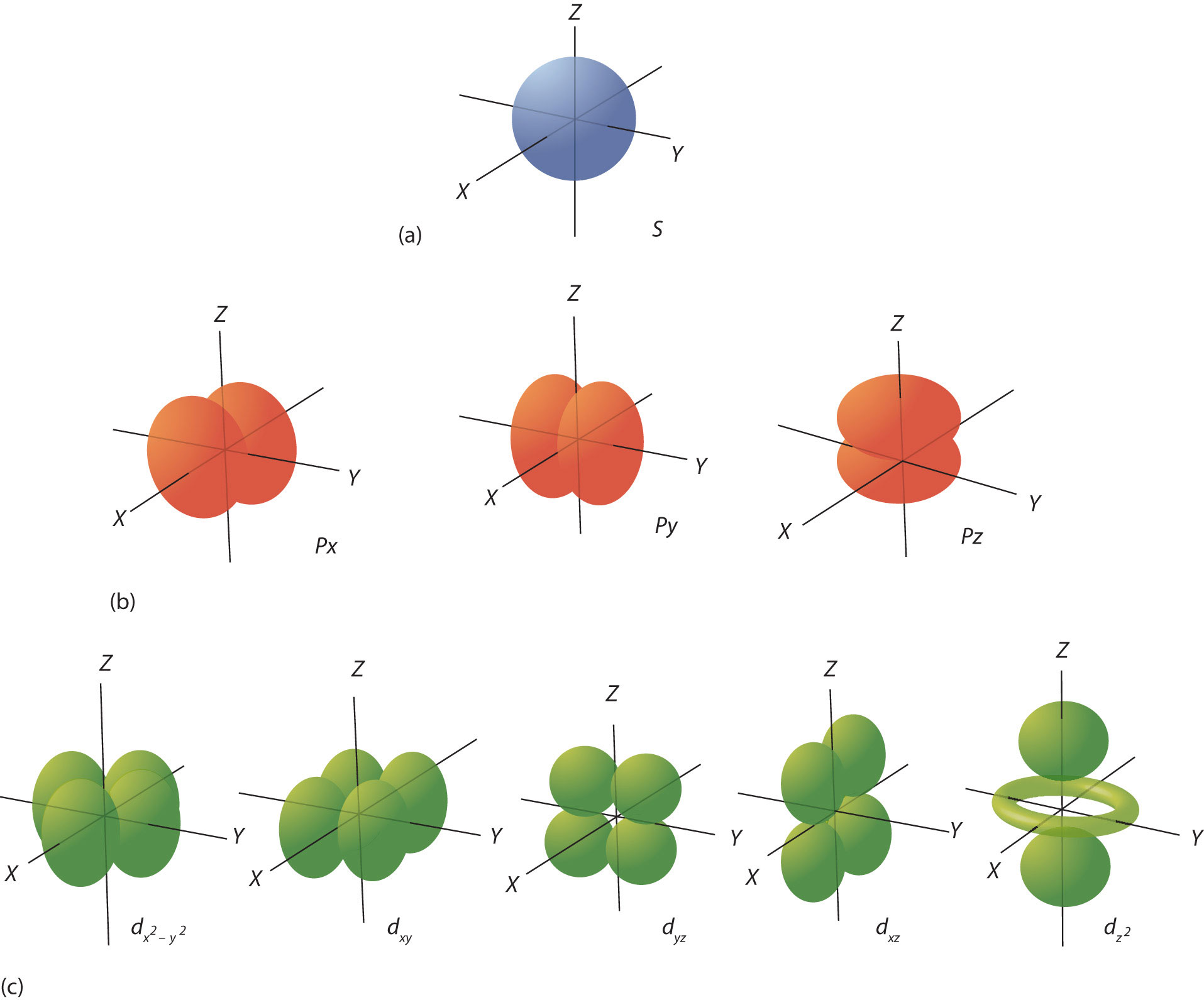
At school, you learn it is discrete shells. But at Uni you learn that each shell contains specific orbitals, each of a unique shape. Each electron pair in the shell occupies its one unique orbital, and they are filled according to the Pauli exclusion principle of lowest energy. The further the shell is from the nucleus, the more unique orbitals it can contain, so the more electrons it can hold - this is because each orbital is described by a standing wave, and the further out the shell is the more nodes it can have so the more complex the orbital patterns can be. At school it was just discrete shells each containing 2,8 etc. (Probably mistaken, but even at uni I don't recall my girlfriend at the time studying phys and molecular chem referring to orbital model (this was 20 years ago thou)?
Orbitals are described as S, P, D or F depending on their overall shape.

[img]
Gassing Station | The Lounge | Top of Page | What's New | My Stuff













