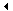Gel filled heat pads. How do they work?
Discussion
Sodium acetate trihydrate crystals have a melting point of 58 °C. When they are heated to around 100 °C, and subsequently allowed to cool, the aqueous solution becomes supersaturated. This solution is capable of supercooling to room temperature without forming crystals. By clicking on a metal disc in the heating pad, a nucleation center is formed which causes the solution to crystallize into solid sodium acetate trihydrate again. The bond-forming process of crystallization is exothermic, hence heat is emitted.
http://www.heatinaclick.com/Products/PocketsSize/t...
I bought three of these last week for a tenner. I was also wondering how they work, thanks for the info!
I bought three of these last week for a tenner. I was also wondering how they work, thanks for the info!
knk said:
Sodium acetate trihydrate crystals have a melting point of 58 °C. When they are heated to around 100 °C, and subsequently allowed to cool, the aqueous solution becomes supersaturated. This solution is capable of supercooling to room temperature without forming crystals. By clicking on a metal disc in the heating pad, a nucleation center is formed which causes the solution to crystallize into solid sodium acetate trihydrate again. The bond-forming process of crystallization is exothermic, hence heat is emitted.
I asked my son, he said " Exotheric reaction to create the heat, ENDothermic reaction to turn it back to the ready state " Apparently he's predicted A* in both Physics and Chemistry at GCSE so I left it at that.
Mr POD said:
knk said:
Sodium acetate trihydrate crystals have a melting point of 58 °C. When they are heated to around 100 °C, and subsequently allowed to cool, the aqueous solution becomes supersaturated. This solution is capable of supercooling to room temperature without forming crystals. By clicking on a metal disc in the heating pad, a nucleation center is formed which causes the solution to crystallize into solid sodium acetate trihydrate again. The bond-forming process of crystallization is exothermic, hence heat is emitted.
I asked my son, he said " Exotheric reaction to create the heat, ENDothermic reaction to turn it back to the ready state " Apparently he's predicted A* in both Physics and Chemistry at GCSE so I left it at that.
Gassing Station | The Pie & Piston Archive | Top of Page | What's New | My Stuff