Discussion
mph1977 said:
'synthetic liquid air' is just a mixture of 21% oxygen and 79% nitrogen - it's slightly safer to work with for chilling stuff on an industrial scale as it;s not an anoxic gas ( like just using liquid Nitrogen) .
ooo, didn't know you could buy that.I've ued liquid nitrogen before to condense liquid oxygen (a coil immersed in liquid N2, blow oxygen throough it from a cylinder, colelct liquid O2 from the other end. It's a really ace blue colour.
You'd be surprised how well digestive biscuits burn if you soak them in liquid oxygen.
RealSquirrels said:
ooo, didn't know you could buy that.
I've ued liquid nitrogen before to condense liquid oxygen (a coil immersed in liquid N2, blow oxygen throough it from a cylinder, colelct liquid O2 from the other end. It's a really ace blue colour.
You'd be surprised how well digestive biscuits burn if you soak them in liquid oxygen.
why didn;t you just get some liquid oxygen ? I've ued liquid nitrogen before to condense liquid oxygen (a coil immersed in liquid N2, blow oxygen throough it from a cylinder, colelct liquid O2 from the other end. It's a really ace blue colour.
You'd be surprised how well digestive biscuits burn if you soak them in liquid oxygen.
or was it a cost thing ? as liquid nitrogen is effectively a 'waste product' of fractional distillation of air iirc. SLA is not a cold as liquid nitrogen but pretty much irrelevant when they use it for quick cooling of frozen food etc ( as the food only needs to go to - 30 c)
Here are phase diagrams for nitrogen, oxygen and CO2
oxygen:

the greek letters are different kinds of crystal forms of solid oxygen, caused by the molecules arranging themselves in different ways at different pressures in the solid state.
nitrogen can't find one.
CO2:
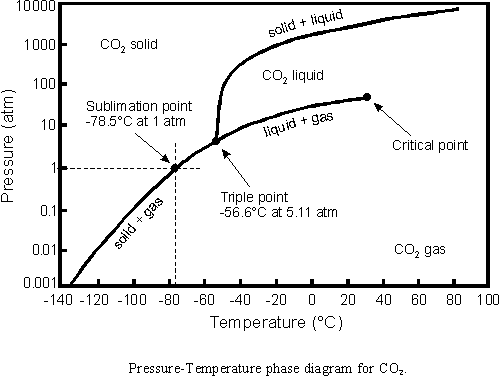
a vapour is not the same as a gas - it's small droplets of liquid suspended in a gas (e.g. air). A gas is a substance in the gaseous state. when you boil water, you get both water vapour (the stuff that we call steam and can see) and gaseous water. of course the two are in equilibium all the time in this case.
anyway, from the phase diagrams, you can see that at a given temperature, if you increase the pressure then a gas will undergo a phase transition to a liquid or solid (sometimes, as with CO2, straight to the solid). So you can compress gases to liquids.
oxygen:

the greek letters are different kinds of crystal forms of solid oxygen, caused by the molecules arranging themselves in different ways at different pressures in the solid state.
nitrogen can't find one.
CO2:

a vapour is not the same as a gas - it's small droplets of liquid suspended in a gas (e.g. air). A gas is a substance in the gaseous state. when you boil water, you get both water vapour (the stuff that we call steam and can see) and gaseous water. of course the two are in equilibium all the time in this case.
anyway, from the phase diagrams, you can see that at a given temperature, if you increase the pressure then a gas will undergo a phase transition to a liquid or solid (sometimes, as with CO2, straight to the solid). So you can compress gases to liquids.

mph1977 said:
RealSquirrels said:
ooo, didn't know you could buy that.
I've ued liquid nitrogen before to condense liquid oxygen (a coil immersed in liquid N2, blow oxygen throough it from a cylinder, colelct liquid O2 from the other end. It's a really ace blue colour.
You'd be surprised how well digestive biscuits burn if you soak them in liquid oxygen.
why didn;t you just get some liquid oxygen ? I've ued liquid nitrogen before to condense liquid oxygen (a coil immersed in liquid N2, blow oxygen throough it from a cylinder, colelct liquid O2 from the other end. It's a really ace blue colour.
You'd be surprised how well digestive biscuits burn if you soak them in liquid oxygen.
or was it a cost thing ? as liquid nitrogen is effectively a 'waste product' of fractional distillation of air iirc. SLA is not a cold as liquid nitrogen but pretty much irrelevant when they use it for quick cooling of frozen food etc ( as the food only needs to go to - 30 c)
RealSquirrels said:
mph1977 said:
RealSquirrels said:
ooo, didn't know you could buy that.
I've ued liquid nitrogen before to condense liquid oxygen (a coil immersed in liquid N2, blow oxygen throough it from a cylinder, colelct liquid O2 from the other end. It's a really ace blue colour.
You'd be surprised how well digestive biscuits burn if you soak them in liquid oxygen.
why didn;t you just get some liquid oxygen ? I've ued liquid nitrogen before to condense liquid oxygen (a coil immersed in liquid N2, blow oxygen throough it from a cylinder, colelct liquid O2 from the other end. It's a really ace blue colour.
You'd be surprised how well digestive biscuits burn if you soak them in liquid oxygen.
or was it a cost thing ? as liquid nitrogen is effectively a 'waste product' of fractional distillation of air iirc. SLA is not a cold as liquid nitrogen but pretty much irrelevant when they use it for quick cooling of frozen food etc ( as the food only needs to go to - 30 c)
BOC have guys who drive tankers of liquid oxygen around all the time, most hospitals have liquid oxygen based systems for their piped oxygen, and my knowledge of the existence of SLA was from a food processing company I had a summer job with in the deep dark past ( and whom my late father was senior engineering projects manager)
Its now used in filters in high end audio equipment if your wallets deep enough you know........ 
http://www.pistonheads.com/gassing/topic.asp?h=0&a...

http://www.pistonheads.com/gassing/topic.asp?h=0&a...
Gassing Station | Science! | Top of Page | What's New | My Stuff


